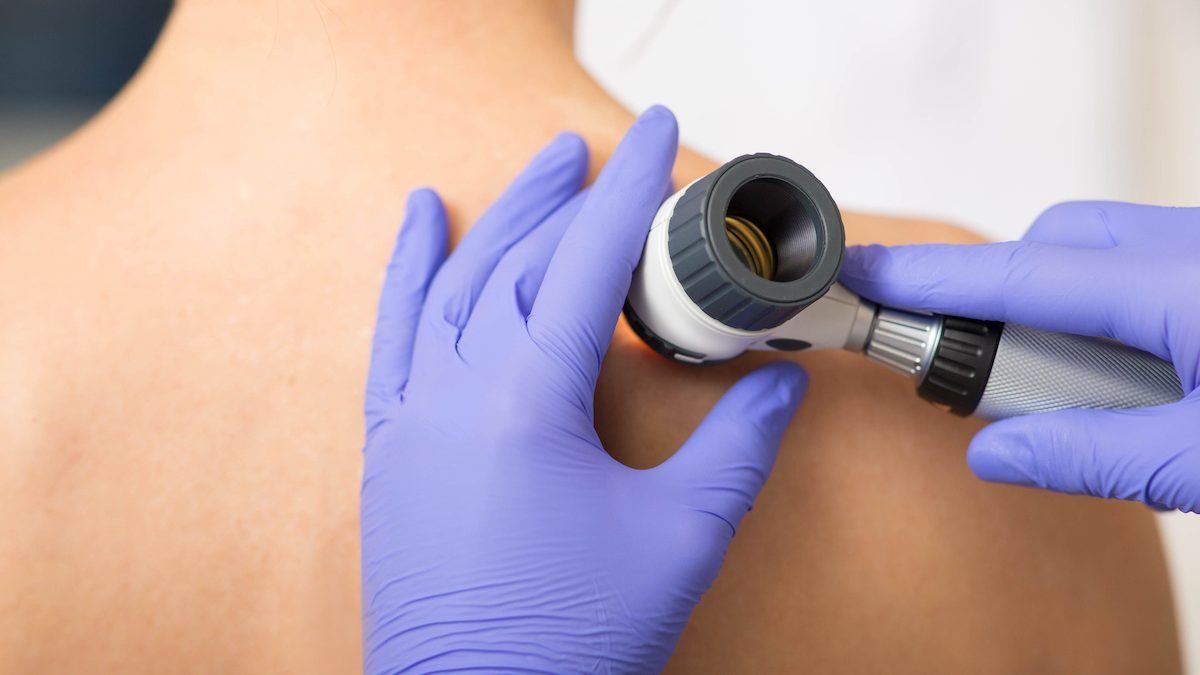
skin cancer: do you know the signs?
skin cancer is the most prevalent form of cancer in canada, accounting for about one-third of all new diagnoses and rates still rising.
we know that the sun causes deadly skin cancer, so why are we still tanning?
the canadian dermatology association has launched a tiktok challenge aimed at a younger demographic to raise awareness of sun safety.
once in the brain, skin cancer cells produce alzheimer's protein: study
the amyloid beta protein that is seen in some cognitive diseases may help cancer spread.
 3 minute read
3 minute read