siblings aged 14 months, 3 and 6 years receive pfizer vax in trials
global study to research outcomes in 4,500 children from four countries
jun 24 2021
 2 minute read
2 minute read


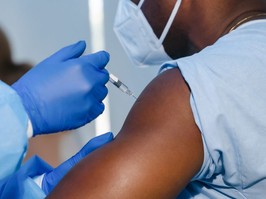
sloan bui, age 14 months, and christian bui, age 3, wait with their mother, erin biro, at the hospital to get their shots.
ochsner hospital for children
by: shari kulha
three young siblings have received their first dose of the pfizer vaccine as part of the company’s global clinical trials for children.
christian bui, 3, and sloan bui, 14 months, were given their first shot on monday at ochsner hospital for children in jefferson, la., a suburb of new orleans, which is one of two hospitals in the state participating in the trials. six-year-old sister ellie bui got her first dose earlier this month, according to an interview the family did with abc news.
the children’s parents, dr. cuong “cj” bui and dr. erin biro, are neurosurgeons at the ochsner medical center.
“for us, our kids living safely in a world where we don’t have to worry about them getting sick from covid, being able to go to school, have playdates with their friends — we feel strongly that vaccine is what is going to get us to those goals,” biro told abc.
“we’re super excited that our entire family now has the opportunity and the chance of possibly being protected,” she added, “and also just contributing to the research and the science to hopefully get all kids across the finish line.”

dr. julia garcia-diaz, ochsner’s director of clinical infectious diseases research, told abc that “a child’s immune system is different than an adult’s immune system, so it is critically important to have a study focused on the efficacy of the vaccine in this cohort.”
advertisement
advertisement
pfizer-biontech, which offered the first covid-19 vaccine to any american over the age of 12, is seeking emergency-use authorization from the food and drug administration to administer its vaccine to younger children by the fall.
the drugmaker began enrolling volunteers in november aged 12 to 17. now, children as young as six months are being enrolled, with ochsner planning on vaccinating up to 75 children. the placebo-controlled study will include up to 4,500 participants from the u.s., spain, poland and finland, according to pfizer.
biro had been increasingly concerned about the so-called delta plus variant, a highly contagious strain of the virus first identified in india in april and now making its way around the world. by june 11, eleven countries had identified a total of 197 patients with the variant,
. britain has seen 36 cases, canada 1, india 8, japan 15, nepal 3, poland 9, portugal 22, russia 1, switzerland 18, turkey 1 and the united states 83. but today, india reported that it had 40 cases in three regions.
“the delta variant has really picked up steam in the u.s. and i think in louisiana especially,” biro told abc news . “given the fact that so much of our population is not vaccinated yet, it has a really significant chance of causing an uptick in covid cases in louisiana.
advertisement
advertisement
“so we feel fortunate that we have the possible chance of having our kids protected, but more so getting the pediatric trial across the finish line so that all kids have the possibility of being protected.”